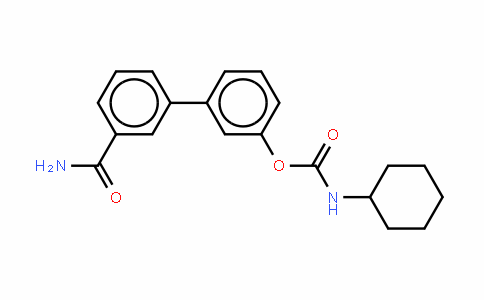
546141-08-6
Chemical Name : URB597,URB-597,KDS4103,KDS-4103/
CAS : 546141-08-6
Synonyms: URB597
Storage:at -20℃ 2 years
Biologieal
URB597 Is Systemically Absorbed after Oral Administration. To determine whether URB597 is absorbed after oral administration, we measured the drug in plasma at various time points after single p.o. dosing in mice (10 or 50 mg/kg). LC/MS/MS analyses of plasma samples taken 15 min after administration revealed that URB597 reached maximal concentrations (Cmax) of 16 ng/ml at the 10 mg/kg dose and 90 ng/ml at the 50 mg/kg dose (Fig. 1A; Table 1). The drug was cleared from circulation within 1 h of administration at the 10 mg/kg dose and within 12 h at the 50 mg/kg dose (Fig. 1A; Table 1). To assess the pharmacodynamic consequences of oral URB597 treatment, in the same set of experiments we determined the ability of this agent to inhibit brain FAAH activity. Ex vivo measurements in brain homogenates showed that URB597 produced a long-lasting inhibition of FAAH activity (Fig. 1B). It is noteworthy that the lower dose of URB597 (10 mg/kg) was cleared more rapidly and produced lower plasma exposure levels (AUC(0–T): 26 ng*h/ml) than did the higher dose (50 mg/kg) (AUC(0–T): 170 ng*h/ml) (Fig. 1A; Table 1). Both doses caused maximal inhibition of FAAH activity (Fig. 1B), albeit with different time courses. The results indicate that URB597 is systemically absorbed and inhibits brain FAAH activity after oral administration.
Reference
The Fatty Acid Amide Hydrolase Inhibitor URB597 (Cyclohexylcarbamic Acid 3′-Carbamoylbiphenyl-3-yl Ester) Reduces Neuropathic Pain after Oral Administration in Mice
-
 Azaphen/
Azaphen/
-
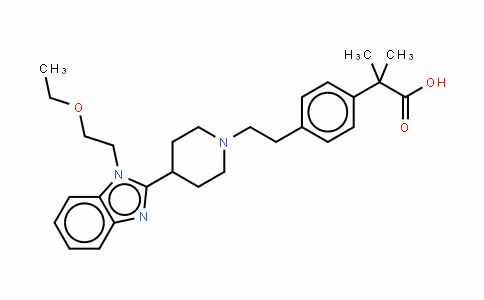 Bilastine/Bilastinum
202189-78-4
Bilastine/Bilastinum
202189-78-4
-
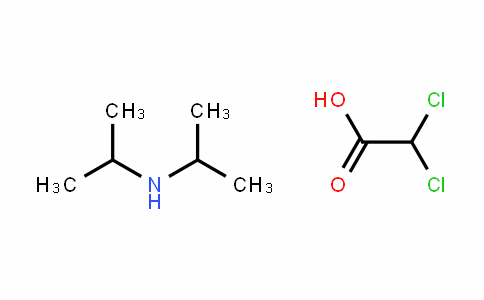 Diisopropylammonium dichloroacetate/
660-27-5
Diisopropylammonium dichloroacetate/
660-27-5
-
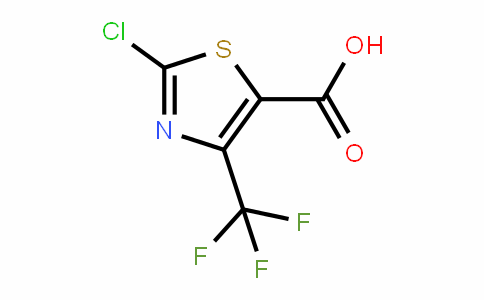 2-CHLORO-4-(TRIFLUOROMETHYL)THIAZOLE-5-CARBOXYLIC ACID
72850-61-4
2-CHLORO-4-(TRIFLUOROMETHYL)THIAZOLE-5-CARBOXYLIC ACID
72850-61-4
 浙公网安备 33010802003999号
浙公网安备 33010802003999号